THREE DECADES, ONE SHOT.
How Dismissed mRNA Went From the Fringes to the Frontline in the Fight Against COVID-19.
January 2020. As the world began murmuring about a mysterious virus emerging from Wuhan, China, the race was already underway at a small biotech firm in Massachusetts. It had been just 48 hours since the SARS-CoV-2 sequence was published and scientists at Moderna had already designed a vaccine that would save countless lives. Not a single viral sample or physical specimen, just digital code.
Within weeks, that digital blueprint was being produced at scale. Within months, it was injected into the arms of millions. To the outside world, it seemed a miraculous, overnight cure almost too efficient to trust. But behind that lightning speed success was a story, three decades in the making. A story of dismissed science, relentless persistence and unwavering belief in a fragile molecule: messenger RNA (mRNA).
The Promise of a Simple Idea
The unlikely hero of this story is mRNA. In the early days of molecular biology, scientists discovered that mRNA acts like the body’s courier system, delivering instructions from DNA to the machinery in cells that makes proteins. Proteins are more than just muscle – they’re enzymes, hormones, immune defenders, and scaffolds that hold your cells together. There are over 100,000 types of proteins at work at any given time, and not a single one of them gets made without mRNA delivering the blueprint.
This understanding sparked a simple but radical idea: if natural mRNA can tell your body what to build, maybe custom-made mRNA could teach cells how to produce certain proteins. In the case of COVID-19, that was the now infamous Spike protein found on the surface of the virus.
Here’s how it worked: the vaccine delivered instructions into muscle cells on how to make harmless fragments of the spike protein. The immune system saw these fragments, sounded its alarm and produced antibodies to defend against what appeared to be an invader. The genius of the approach? When the real virus eventually showed up, the body’s immune system was primed to respond quickly and effectively.
mRNA was first discovered in 1961 but it wasn’t until 1984 that attempts began to bring this idea to life. That year, a group of researchers from Harvard – Paul Krieg, Doug Melton, Tom Maniatis and others – managed to produce biologically active mRNA in the lab, and it worked just like the natural stuff, it produced proteins!
But enthusiasm was cautious. “RNA in general had a reputation for unbelievable instability”, recalled Paul Krieg in a 2021 Nature retrospective¹ , “Everything around RNA was cloaked in caution”. Rather than patent the technology, the team handed over their reagents to Promega, a lab supplies company, in exchange for some modest royalties and famously, a few cases of Veuve Clicquot champagne!
From here, researchers like Robert Malone picked up the baton. He looked into wrapping up the synthetic mRNA into tiny fat bubbles called liposomes to shield it from degradation and help it enter cells more efficiently. These experiments showed hopeful signs, but obstacles loomed. mRNA was incredibly fragile, breaking down quickly once in the body, and worse, the immune system recognised it as a viral invader sparking inflammation. Delivering mRNA safely was like trying to drop a message behind enemy lines – hard to keep intact without triggering alarm bells. Malone’s lipid-encapsulation work later became a point of contention, as he claimed a central role in the mRNA vaccine development, a claim that has sparked ongoing debate.
As the biotech boom focused its attention on more promising candidates like DNA therapies and protein drugs, mRNA began losing credibility and momentum. Grants dried up. Interest moved on.
The Foundations
In 1995, a determined Hungarian biochemist named Katalin Karikó sat in her underfunded lab at the University of Pennsylvania, staring down at yet another rejection letter. Her grant application was a plea to secure support for her work on synthetic mRNA. But it failed, again, and this time it was accompanied by a demotion.
Her focus on mRNA faced repeated rejection in the scientific community. Colleagues questioned its viability, and grant agencies offered little support. Yet Karikó remained committed, sacrificing promotions and paychecks to keep her work alive. Then, in one of sciences’ now notorious partnerships, Karikó met Drew Weissman, a UPenn immunologist interested in developing vaccines. They began collaborating, driven by a shared belief that mRNA had a future worth pursuing.

Katalin Karikó, Ph.D., and Drew Weissman, M.D., Ph.D.
Their biggest challenge was the violent immune response evoked upon injection of mRNA into cells. It was like the body thought itself to be under viral siege. For five years they searched for a way to get around these immune defenses. Then, in 2005, they had a ‘eureka moment’ discovery. By swapping one of mRNA’s four chemical bases, uridine (U), with a subtly modified version called pseudouridine (ψ), they could sneak the molecule past the immune system. The modified RNA was no longer seen as a threat. It slipped into cells, delivered its message, and did its job without aggravating the immune system.
They had solved one of the core problems of mRNA therapy. But when they published their findings, the science world barely even batted an eye and their paper garnered few citations. The university couldn’t see the breakthrough’s potential and sold the patent rights for a modest sum. But Karikó and Weissman weren’t in it for the credit. They kept refining the technique, improving the purity of the mRNA, and demonstrating that it worked again and again. In the dim corners of academia, while others chased more glamorous fields, they laid the groundwork for what would one day underpin a life-saving vaccine.
Yet their work was only one patch of a much larger tapestry. The eventual success of mRNA vaccines depended on a constellation of innovations: lipid nanoparticle delivery systems to protect and transport the fragile molecule, sophisticated purification techniques, improved thermostability, and large-scale manufacturing processes. These parallel advances converged to make clinical-grade mRNA vaccines a reality.
Betting on a Long Shot
Karikó and Weissmann were not alone in their ‘delusions’. In Germany, a similar misfit husband-and-wife duo Ürgür Sahin and Özlem Türeci founded BioNTech in 2008. They set out with a bold vision to use mRNA to train the immune system against cancer. It was an unconventional path that was going to require meticulous science and long-term investment. Cancer’s complexity and mRNA’s fragility made the pairing seem implausible to most researchers. However, slowly and methodically they refined mRNA delivery systems, studied immune responses, and published early data.
Meanwhile in the U.S., another academic outsider was taking note. In 2010, a Harvard stem cell biologist named Derrick Rossi used modified mRNA to reprogram adult stem cells, a breakthrough that landed him in TIME magazine’s list of ‘The Year’s Most Influential People’. Though Rossi wasn’t business-minded, his discovery drew attention from some savvy investors. With backing from Flagship Ventures and a team of co-founders, Rossi helped launch a company that would later be known worldwide as Moderna.
Both BioNTech and Moderna were, in a sense, spiritual heirs to the early work of many unsung researchers. Without that proof-of-concept, neither company would have had a viable platform to bounce off. Their success, like all in biopharma, did not emerge in a vacuum, instead, it was built through two decades of dogged persistence in academic labs, often overlooked and underfunded.
By 2019, both companies were ready. They had advanced mRNA platforms, clinical trial data in hand, and manufacturing infrastructure in place. But the world only viewed mRNA as a now-promising science, not a practical solution. They were companies in the long grass, waiting.
Then came the virus.
When Preparation Became Urgency
When Chinese scientists published the SARS-CoV-2 genome in January 2020, Uğur Sahn at BioNTech read it and immediately sensed what others didn’t: this signalled the start of a global pandemic. Within days, he made the call to pivot BioNTech away from its long-standing cancer immunotherapy research and toward a COVID-19 vaccine. Almost in unison, across the pond, scientists at Moderna reached the same conclusion. With decades of groundwork already laid, both companies were uniquely positioned to respond.
Within a mere 12 weeks, clinical trials were under way. Moderna’s Phase I trial launched just 66 days after the viral genome was published. BioNTech partnered with Pfizer and followed close behind. When late-stage trials returned efficacy rates above 94%, the scale and speed of what had been achieved became undeniable. While the mRNA vaccines took center stage, they weren’t the only contenders. Other platforms, like adenoviral vector vaccines, were being used by AstraZeneca and Johnson & Johnson. These alternatives were more stable and easier to store, making them attractive for global distribution. But mRNA had one major advantage: adaptability. Scientists could rapidly update the vaccine blueprint to match emerging variants. So if the virus mutated, new versions could be developed within weeks. In a fast-moving pandemic, that kind of speed changed everything.
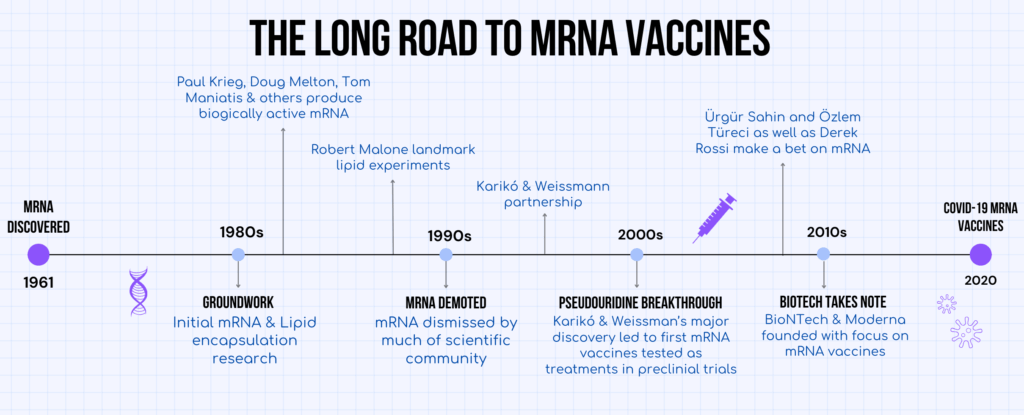
Infographic of mRNA’s timeline. Made using Canva
By December, emergency use authorisations were granted and the first doses were being administered to healthcare workers and vulnerable populations around the world. This compressed timeline, which would usually take a decade, was unprecedented. Yet outside the labs, the vaccines’ reception was clouded by hesitation. No other medication with a development arc so long had reached the public with such momentum. Ironically, the urgency that had enabled this rapid coordination, funding, and approval was what made many question its validity.
The mRNA vaccine rollout wasn’t just a public health challenge, it was a communication challenge. The scientific community had to convince the public that what looked like a sprint was actually the final few meters of a marathon no one had noticed until the finish line. But hesitancy didn’t stem from speed alone. It was rooted in deeper social undercurrents: decades of medical mistrust, compounded by political polarisation and a flood of misinformation amplified by social media. It was a reminder that scientific trust isn’t earned in press releases or peer-reviewed journals alone. It’s forged, and sometimes fractured, in the complicated spaces where health, history, and identity overlap.
Recognition, Resentment and the Race for Credit
The world watched as mRNA vaccines helped curb a deadly pandemic. For many, it was a triumph of modern science. Proof that decades of molecular biology and biotech investment could finally pay off in real-time. When Katalin Karikó and Drew Weissman were awarded the Nobel Prize in Physiology or Medicine in 2023, it was a deeply symbolic moment. Two scientists who had once toiled in obscurity, dismissed and demoted, were now receiving the highest honour of recognition for work that had saved millions of lives.
But in the shadows of celebration, contestants of credit and recognition lingered. Robert Malone, who had conducted early mRNA-lipid experiments, stepped into the spotlight to argue that he had been unfairly written out of the vaccine’s origin story. His early work was thrust back into the spotlight, along with debates over the selective memory of scientific institutions. Who gets cited, credited, or canonised often has less to do with scientific merit and more to do with timing, visibility, and institutional power. His case is a window into a broader, uncomfortable truth—many scientific contributions are orphaned by the systems meant to recognise them.
In commercial biotech, tensions also simmered. Moderna filed a lawsuit against Pfizer and BioNTech². BioNTech, in turn, faced legal action from CureVac, another mRNA pioneer, over patents related to sequence design³. Legal action questioned not just patents, but pride. Who had truly pioneered what? And who got to put their name on molding this legacy?
What emerged wasn’t just academic squabbles or a scramble for royalties. It was a human story about validation, visibility, and the uneasy marriage between scientific progress and recognition. Each company and researcher had contributed in their own way. The technology had gone from academic orphan to industrial goldmine, and now, everyone wanted to be remembered— not just for what they built, but for daring to believe in it first.
Moral of the Story
While this story can be told as a neat arc from idea to implementation, in reality, it unfolded in fits and starts, with many conflicting claims, forgotten contributions, and missed opportunities. The story of mRNA isn’t just about molecules and patents, it’s about persistence in the face of indifference, and the quiet determination to keep going when no one is watching.
Karikó, whose ideas were dismissed and career demoted, chose resilience over recognition. Sahin and Türeci, quietly building in Germany, ignored trends to follow their conviction. Moderna, once scoffed at, became the torchbearer of a technology few had believed in. And Robert Malone, whose early experiments were ahead of their time, rose difficult but necessary questions about recognition, memory, and the politics of scientific credit. None of these breakthroughs belonged to one person or moment. They were the product of a community of believers, bound not by certainty but by the sense that the science mattered.
When the pandemic arrived, the world was unprepared, but mRNA wasn’t. The people who had devoted years, even decades, to this fragile technology, without reward or recognition, became the unsung architects of the world’s defense. Their persistence, once dismissed as misplaced, was suddenly the reason the world could respond at all. Millions of lives were saved not by accident or haste, but by a commitment sustained long before the crisis came. Yet even in that moment of arrival, their work was met with a complicated reception of hope, but hesitation. To many, the vaccine seemed to appear too quickly to be trusted. But that is the irony: the thing that saved us was built precisely because its champions didn’t wait for permission, or applause.
This wasn’t just a story of scientific ingenuity. It was a story of emotional stamina, of people who endured scepticism, failure, and obscurity. Who bet on a fragile molecule and the promise it carried. Their story is a reminder that the most transformative work often happens far from the spotlight, by people willing to believe in what the world doesn’t yet understand.
Three decades, one shot. The world wasn’t ready but the science was.
¹ Callaway, Ewen. The tangled history of mRNA vaccines. Nature 597, 318–324 (2021).
² Sandle, Paul. “Moderna secures partial victory in COVID vaccine legal fight.” Financial Times, June 2025.
³ Reuters Staff. “Pfizer convinces US Patent Office to cancel two Moderna COVID-19 vaccine patents.” Reuters, March 2025.